Introduction: Why Concept Comes Before Calculation
If you’re sitting in AP Chemistry class or flipping through past exams, you’ve probably noticed a pattern: the questions that feel the simplest on paper often demand the deepest conceptual understanding. Equilibrium and reaction rates are a perfect example. Too many students treat these topics as formula farms — memorize an expression here, plug-and-chug there — and then feel blindsided when a question asks for explanation, prediction, or qualitative reasoning.
This blog is your guided tour of the ideas that matter most. We’ll build intuition for what equilibrium and rates really mean, compare the two so you see their relationship, and show how to translate qualitative understanding into correct calculations when you need to. Along the way you’ll get study strategies, example problems explained conceptually, and a suggested study schedule. If you want extra help, Sparkl’s personalized tutoring (1-on-1 guidance, tailored study plans, expert tutors, and AI-driven insights) can fit naturally into the plan — but only when you want targeted support.

Part 1 — Big Picture: What Equilibrium and Rates Tell Us
Equilibrium: A Dynamic Balance
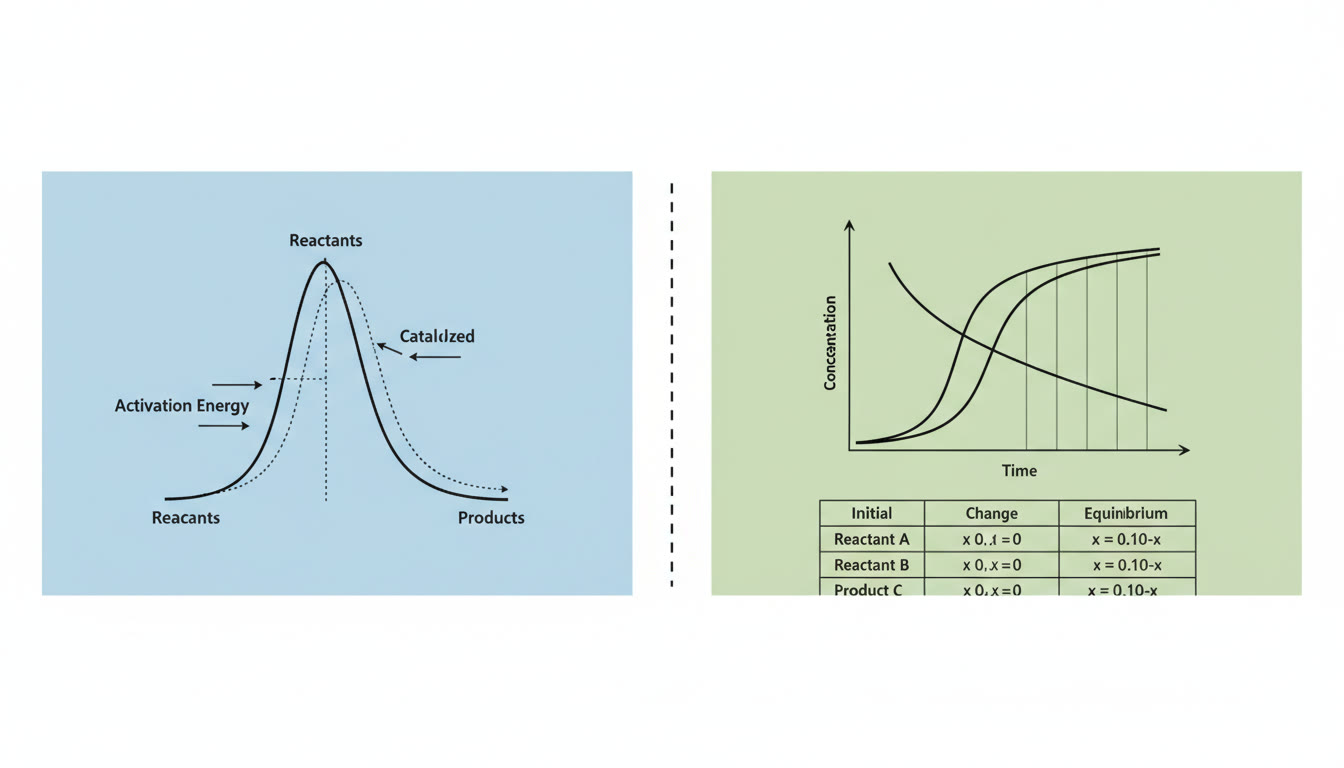
Equilibrium is not a standstill; it’s a balance of opposing processes. In a reversible reaction A ⇌ B, molecules keep reacting forward and backward, but at equilibrium the rates of the forward and reverse reactions are equal. The concentrations may be constant, but molecules are still moving and colliding.
Key concept: equilibrium describes what happens when the reaction system has no net macroscopic change in concentration because opposing microscopic events cancel out. The equilibrium constant (K) summarizes the ratio of product to reactant activities at that balance point, but K doesn’t tell you how fast the system gets there.
Reaction Rates: The Pace of Change
Rates describe how quickly a reaction proceeds — the speed at which reactants are consumed and products formed. Rate laws (rate = k [A]^m [B]^n) capture how concentration affects speed, while the rate constant k encodes temperature and activation energy information. Conceptually, kinetics is about the path a reaction follows; equilibrium is about the destination.
Key concept: kinetics answers “How fast?” while thermodynamics (equilibrium) answers “Where will it settle?” Both are essential, but they give different types of information.
How the Two Fit Together
Imagine you’re climbing a hill to a plateau. Kinetics determines how fast you reach the plateau (rate) and whether you even have the energy to climb the hill (activation energy); equilibrium determines the plateau’s height relative to the valley — i.e., where the balance lies. A reaction can be thermodynamically favorable (products favored at equilibrium) but kinetically slow (takes a long time to get there) — or vice versa.
Part 2 — Building Intuition: Qualitative Reasoning That Works on the Exam
Le Châtelier’s Principle Without the Jargon
Le Châtelier’s Principle is an elegant, testable idea: if you disturb a system at equilibrium, the system shifts to partially oppose the disturbance. But instead of rote lists, think in terms of cause-and-effect.
- If you add a reactant, the forward reaction has more material to consume, so the system shifts toward products.
- If you remove a product, the system makes more product to replace what was lost.
- If you increase pressure by decreasing volume in a gas-phase system, the equilibrium shifts to the side with fewer moles of gas.
- If you heat an endothermic reaction, the system shifts toward products; if you heat an exothermic reaction, it shifts toward reactants.
Practice question you can do in your head: For N2O4(g) ⇌ 2 NO2(g), what happens when you increase pressure? Think: pressure up → volume down → favors side with fewer gas molecules → shift left toward N2O4.
Rates Through the Collision Lens
Picture molecules as tiny billiard balls: increasing temperature increases molecular speed, which raises the number of collisions and the fraction of collisions energetic enough to overcome the activation energy. Changing concentration increases collision frequency similarly.
Use these quick checks: raise temperature → rate increases; add catalyst → activation energy lowers → rate increases but equilibrium constant (K) remains the same; change concentration → initial rates are affected but K is unchanged.
Common Misconceptions to Avoid
- Equilibrium means equal concentrations. False. Equal rates, not concentrations. K determines relative concentrations.
- K depends on how quickly the reaction occurs. False. K depends only on temperature (and reaction identity), not on rate constants directly — though there is a relation via ratio of forward and reverse rate constants at the molecular level.
- Catalysts change equilibrium. False. Catalysts speed up both forward and reverse pathways equally, reaching equilibrium faster without changing K.
Part 3 — Concept-First Examples (Translate Ideas Into Results)
Example 1: Predicting Shift From a Disturbance
Scenario: Consider the equilibrium system H2(g) + I2(g) ⇌ 2 HI(g). If a small amount of H2 is suddenly added, what happens to the concentration of HI and why?
Conceptual answer: Adding H2 increases the forward reaction’s reactant pool, temporarily increasing the forward rate. The system responds by producing more HI until the forward and reverse rates match again at a new equilibrium with higher [HI]. No calculations required: follow which rate is immediately affected and let the system’s response follow.
Example 2: Temperature and Rates vs. Equilibrium
Scenario: For an exothermic reaction, heating the system increases kinetic energy. What happens to the rate and to the position of equilibrium?
Conceptual answer: Heating generally increases rates (molecules move faster), so both forward and reverse reactions speed up. But for an exothermic reaction, adding heat is like adding a product; Le Châtelier’s principle predicts a shift toward reactants, changing the final equilibrium composition even though both reactions are faster.
Part 4 — Turning Conceptual Insight Into Calculations
Stepwise Approach to Equilibrium Problems
Before you write an ICE table, follow these mental steps:
- Identify the balanced reaction and whether K refers to concentrations (Kc) or partial pressures (Kp).
- Ask: Is temperature changing? If yes, K might change; if not, K is constant.
- Decide which disturbance (if any) was applied: added/removed substance, volume/pressure change, temperature change. Predict qualitatively first.
- Only after prediction, set up the ICE table and solve for concentrations. Use approximations (x<<initial) carefully and check them numerically afterward.
That last step is important: a clear conceptual prediction helps you choose whether approximations are safe and guides your algebra.
Example 3: ICE Table Guided by Concept
Problem sketch: Given N2O4 ⇌ 2 NO2 with Kc = 0.15 at a certain temperature. You start with pure N2O4 at 0.200 M. What are equilibrium concentrations?
Concept-first: You know forward reaction produces two moles of NO2, so if x moles of N2O4 decompose, NO2 forms 2x. Set up ICE with forward change -x, +2x. Solve the algebra (omitted here) — while solving, if x is small relative to 0.200, you can approximate. Always check the assumption numerically at the end.
How K and Rate Constants Relate (Without Getting Lost in Algebra)
At a mechanistic level, for a simple reversible elementary reaction A ⇌ B, the equilibrium constant K equals k_forward / k_reverse. That’s a precise relation for elementary steps. Conceptually, K captures the ratio of how easily the system goes forward vs. backward, while the magnitudes of the rate constants and activation energies control how fast those processes happen. In multi-step mechanisms, the relation between observable rate laws and K becomes more nuanced, but the conceptual link remains.
Part 5 — Practical Study Strategies: Concept First, Practice Second
Daily 4-Phase Routine (30–60 minutes)
- Warm-up (5–10 min): Quick conceptual flashcards — e.g., “What does K change with?” “How does a catalyst affect equilibrium?”
- Deep Read (10–15 min): One short section of your textbook or AP Classroom notes — focus on verbal explanations and diagrams, not algebra.
- Mental Prediction (10–15 min): Look at examples or short prompts and predict outcomes qualitatively before calculating.
- Practice Problems (10–20 min): Do 2–3 problems that require both reasoning and calculation; after solving, explain your steps out loud or in writing emphasizing the conceptual triggers.
Why this works: frequent, bite-sized conceptual practice primes your intuition so the exam’s trickier wording becomes manageable. When you build an “instinct” for what should happen, calculations become mechanical checks — not the source of your answer.
Weekly Deep-Dive: Lab, Mechanisms, and Error Analysis
Once per week, spend one longer session (1.5–2 hours) on hands-on or virtual lab experiments, mechanism mapping, and error analysis. Relating experiments to equilibrium shifts (e.g., observing color changes in equilibrium mixtures) cements understanding. Also practice explaining why a common student mistake is wrong — that technique prepares you for FRQ-style reasoning tasks.
Part 6 — Exam-Focused Tips and Time Management
Multiple-Choice Strategies
- Read the question and immediately ask: Is this testing kinetics, equilibrium, or both? A phrase like “after 5 minutes” often suggests kinetics.
- If it’s a disturbance problem, predict qualitatively first; this often eliminates 2–3 answer choices quickly.
- For rate law questions, be skeptical of stoichiometry-based assumptions; rate laws are determined experimentally unless the step is explicitly labeled elementary.
Free-Response Strategies
In FRQs, the graders are looking for logical reasoning as much as correct numbers. Structure your answers:
- State the concept (e.g., Le Châtelier’s Principle) in one sentence.
- Explain the immediate effect on rates (qualitative).
- Predict the direction of shift and explain why, linking to concentrations or partial pressures.
- Do the calculation, but circle your conceptual conclusion — graders often award points for correct reasoning even if arithmetic is off.
Part 7 — A Simple Study Plan (6 Weeks Before the Exam)
| Week | Focus | Daily Goal |
|---|---|---|
| 1 | Foundations: Collision Theory, Rate Laws, Intro to Equilibrium | Concept flashcards + 2 practice problems |
| 2 | Kinetic Mechanisms and Rate-Determining Steps | Sketch mechanisms + solve one mechanism-based rate law problem |
| 3 | ICE Tables and K Calculations | 2 ICE tables and conceptual write-ups |
| 4 | Le Châtelier’s Principle and Thermodynamic Links | Predict shifts for 5 scenarios and justify |
| 5 | Full Mixed Practice: Timed MC sets + 1 FRQ | Timed section practice + FRQ planning |
| 6 | Review Weak Spots, Simulated Exam Session | One full practice exam section; targeted review |
Notice how the plan always starts qualitatively and only later layers in calculation-heavy practice. This mirrors how a chemist thinks: explain the scene, then quantify.
Part 8 — Sample Problem Walkthrough (Concept First, Then Algebra)
Problem (Sketch)
At 350 K, the equilibrium for Hb ⇌ 2 C has Kc = 0.50. If initial [Hb] = 0.400 M and [C] = 0, what is [C] at equilibrium?
Conceptual roadmap
Predict direction: starting with only reactant Hb, the forward reaction will produce C until equilibrium. Since K is less than 1, the equilibrium favors reactants, so we expect a modest formation of C, not complete conversion.
ICE setup (concise)
Let x = mol Hb reacted. Then [Hb]eq = 0.400 – x, [C]eq = 2x. Plug into Kc expression (2x)^2 / (0.400 – x) = 0.50. Solve for x, check approximation x << 0.400 if used, and interpret the result in context of the initial conceptual prediction.
Notice how the qualitative expectation (K < 1 → reactant favored) gives you a check on whether the numeric root makes sense.
Part 9 — How to Use Tutoring Effectively (Including a Note About Sparkl)
Tutoring can accelerate your conceptual mastery — but not all tutoring is equal. The most effective sessions focus on:
- Diagnosis: pin down which conceptual leaps are shaky (e.g., confusing rate vs. equilibrium).
- Active practice: the tutor makes you predict, explain, and teach back problems.
- Personalized pacing: more time on your weaker topics and targeted FRQ practice.
If you’re considering help, Sparkl’s personalized tutoring offers 1-on-1 guidance, tailored study plans, expert tutors, and AI-driven insights that can identify patterns in your mistakes and recommend focused practice. Used sparingly and intentionally, tutoring helps turn conceptual confusion into clear, test-ready intuition.
Part 10 — Final Checklist Before Test Day
- Be able to explain in one sentence the difference between kinetics and equilibrium.
- Practice making quick qualitative predictions for shifts and justify them with Le Châtelier’s reasoning.
- At least once, do a timed section where you force yourself to predict first, calculate second.
- Know how to set up ICE tables and when approximations are valid; always check numerically.
- Be familiar with typical traps: assuming stoichiometric coefficients equal rate orders, or thinking catalysts change K.
Conclusion: Think Like a Chemist, Not a Calculator
Equilibrium and reaction rates are less about memorizing many disjoint formulas and more about building a way of seeing chemical systems. Train your intuition: ask what the immediate effect of a disturbance is on rates, what direction the system will shift, and how temperature or catalysts will affect pace versus position. Calculations become your verification tools — they confirm what your conceptual thinking predicted.
When you practice this approach, you’ll find the AP exam’s trickiest questions stop being traps and start being opportunities to show clear scientific thinking. If you want targeted help building that intuition faster, consider short blocks of personalized tutoring — for example, Sparkl’s expert tutors can design one-on-one sessions and tailored study plans to strengthen exactly the areas where you need conceptual clarity.
Above all: stay curious. Chemistry rewards students who think in stories and relationships. Equip yourself with the habit of predicting, explaining, and then calculating — and the scores will follow.














No Comments
Leave a comment Cancel