Why Kinetics Matters (and Why You Should Care)
Chemical kinetics — the study of how fast reactions occur and why — is the part of AP Chemistry that turns equations into stories. Instead of asking what reacts with what, kinetics asks: how quickly? What steps make the reaction happen? How can we speed it up or slow it down? For students, mastering kinetics strengthens problem-solving, deepens conceptual understanding, and delivers big wins on free-response and multiple-choice sections.

Big Picture: Rate Laws, Mechanisms, and Catalysts
Think of a chemical reaction as a play. The rate law is the script that tells you what controls the pace. The reaction mechanism is the sequence of scenes — the elementary steps — that create the full performance. Catalysts are guest directors who rearrange the scenes to make the play faster (or occasionally more selective), without appearing in the final curtain call.
Understanding the interplay between these three concepts — rate laws, mechanisms, and catalysts — lets you predict, interpret, and manipulate reactions in both lab and exam settings.
Key Definitions (Quick Reference)
- Rate: Change in concentration per unit time (e.g., M s−1).
- Rate Law: Expression linking rate to reactant concentrations, often rate = k[A]^m[B]^n.
- Rate Constant (k): Proportionality constant that depends on temperature and activation energy.
- Order of Reaction: The exponent m or n (or overall order = m + n) that describes how rate depends on concentration.
- Reaction Mechanism: A sequence of elementary steps that produce the overall reaction; the slowest elementary step is the rate-determining step (RDS).
- Catalyst: Substance that increases reaction rate by lowering activation energy or enabling alternative pathways, and is regenerated by the end of the reaction.
Rate Laws: How to Read the Script
The rate law is never deduced from the balanced overall equation alone. Instead, you must rely on experimental data or a known mechanism. Here’s the general form you’ll see on AP: rate = k[A]^x[B]^y. The exponents x and y come from experiments — they tell you how sensitively the rate responds to concentration changes.
Common Orders and What They Mean
- Zero Order: rate = k. The rate is constant and independent of reactant concentration (rare but seen with surface-catalyzed reactions).
- First Order: rate = k[A]. Doubling [A] doubles the rate. Radioactive decay and many simple unimolecular processes follow first-order kinetics.
- Second Order: rate = k[A]^2 or rate = k[A][B]. Doubling concentrations can quadruple the rate, depending on the form.
Interpreting experimental data — initial rates, integrated rate laws, or half-life analyses — is a core AP skill. Practice translating tables of concentration and rate into the corresponding rate law and constant.
Worked Example: Finding a Rate Law from Initial Rates
Suppose experimental data show:
| Experiment | [A] (M) | [B] (M) | Initial Rate (M s−1) |
|---|---|---|---|
| 1 | 0.10 | 0.10 | 2.0 × 10−4 |
| 2 | 0.20 | 0.10 | 4.0 × 10−4 |
| 3 | 0.10 | 0.20 | 2.0 × 10−4 |
Compare experiments 1 and 2: [A] doubles while [B] stays fixed; rate doubles, so rate ∝ [A]^1. Now compare 1 and 3: [B] doubles while [A] stays the same; rate stays the same, so rate ∝ [B]^0. Thus rate = k[A]. The reaction is first order in A and zero order in B (overall first order).
Integrated Rate Laws and Half-Lives — What AP Loves
Beyond initial rates, you’ll sometimes be asked to identify the order using concentration vs. time plots or to calculate half-lives. Memorize the three integrated forms and their graphical signatures:
- Zero order: [A] vs. time is linear. slope = −k. Half-life depends on [A]0.
- First order: ln[A] vs. time is linear. slope = −k. Half-life is constant (t1/2 = ln2 / k).
- Second order: 1/[A] vs. time is linear. slope = k.
Tip: If you see a straight line for ln[A] vs. time, think first order immediately — that saves time during the exam.
Mechanisms: Assembling the Scenes
The mechanism explains the rate law. For multi-step reactions, only an elementary step’s molecularity directly relates to concentration dependencies: unimolecular (A → products) leads to first-order behavior, bimolecular (A + B → products or A + A → products) leads to second-order behavior in the participating reactants, and so on.
Rate-Determining Step (RDS)
The slowest elementary step controls the rate law. If the RDS involves only species explicitly present in the elementary step, the rate law follows that molecularity. But if the RDS includes an intermediate, or if a fast pre-equilibrium precedes it, you’ll often need to express intermediate concentrations in terms of reactants using equilibrium expressions.
Example Mechanism and Deduction
Consider the overall reaction: A + B → products. Suppose the proposed mechanism is:
- Step 1 (fast equilibrium): A + B ⇌ I (intermediate) (K = [I]/([A][B]))
- Step 2 (slow): I → products
If Step 2 is slow and rate = k2[I], substitute [I] from Step 1: [I] = K[A][B], so rate = k2K[A][B], which yields rate ∝ [A][B] — second order overall, first order in each reactant. This kind of reasoning frequently shows up on AP free-response questions.
Catalysts: The Invisible Helpers
Catalysts provide alternate pathways with lower activation energies and often involve transient intermediates that are regenerated. They do not change the overall thermodynamics (ΔG, ΔH) but affect kinetics by changing the activation energy and the reaction coordinate.
Common Types of Catalysis
- Homogeneous Catalysis: Catalyst is in the same phase as reactants (e.g., acid-catalyzed esterification in solution).
- Heterogeneous Catalysis: Catalyst is in a different phase (e.g., gas reactions over a metal surface like in catalytic converters).
- Enzymatic Catalysis: Biological catalysts are highly specific and often show Michaelis–Menten kinetics (this can be extended on AP but focus on basics: lower Ea, better orientation, transition state stabilization).
In AP problems, catalysts may appear in reaction mechanisms where they form transient complexes and are regenerated at the end of the mechanism. Always track the catalyst in mechanism steps; it should be present on both sides of the overall mechanism but not in the net stoichiometry if everything cancels properly.
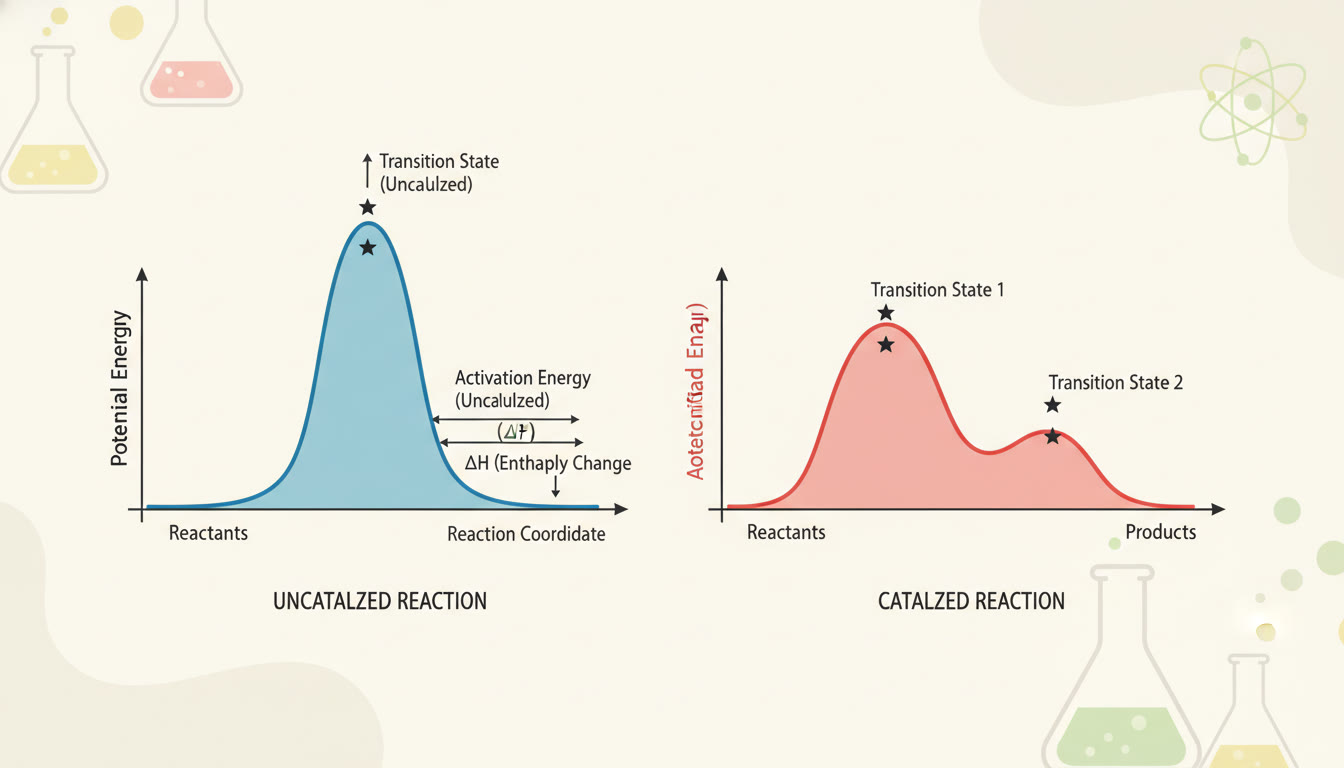
Animating Kinetics: Energy Diagrams and the Arrhenius Equation
Energy diagrams show potential energy vs. reaction coordinate. The difference between reactant energy and transition state energy is the activation energy (Ea). The Arrhenius equation links k to Ea and temperature (T): k = A e^(−Ea/RT). Practically, this tells you that even small increases in temperature can significantly increase rate constants for reactions with large Ea.
AP-Relevant Takeaways
- Lower Ea (via catalysts or different mechanisms) → larger k → faster reaction.
- Higher temperature → larger k, often examined qualitatively or with students asked to calculate changes using Arrhenius or comparative reasoning.
- Activation energy is an intrinsic barrier; catalysts change the path but not the reactants’ or products’ energies.
Useful Problem-Solving Strategies for AP
Kinetics questions are logic puzzles. Here are practical strategies that consistently work on AP exams and in studying:
- Identify what’s given: Are you working with initial rates, concentration vs. time data, or mechanism steps?
- Start from experiments: Use ratios (Experiment A/Experiment B) to isolate orders.
- Use dimensions: Units can hint at overall order: k units change with order (e.g., M s−1 for first order? check!).
- Translate mechanism to math: For elementary steps, molecularity → exponent in rate law; for multi-step, find the RDS and substitute intermediates when necessary.
- Sketch energy diagrams: Visuals help you argue about catalysts and transition states on free-response parts.
Table: Quick Reference for Orders and Signatures
| Order | Rate Law | Integrated Form | Graphical Signature | Half-Life Dependence |
|---|---|---|---|---|
| Zero | rate = k | [A] = [A]0 − kt | [A] vs. t linear | Depends on [A]0 (not constant) |
| First | rate = k[A] | ln[A] = ln[A]0 − kt | ln[A] vs. t linear | Constant (t1/2 = ln2/k) |
| Second | rate = k[A]^2 or k[A][B] | 1/[A] = 1/[A]0 + kt | 1/[A] vs. t linear | Depends on [A]0 |
Exam-Ready Examples and Common Traps
Example 1: You’re given a mechanism where Step 1 is slow and involves two molecules colliding — A + B → C (slow); Step 2 is fast. The RDS is bimolecular, so you might conclude rate ∝ [A][B]. But watch for intermediates or pre-equilibria that change the math.
Trap: Assuming stoichiometric coefficients equal orders. The balanced equation tells you nothing about the rate law unless it’s an elementary reaction.
How To Answer Free-Response Kinetics Questions
- State assumptions clearly (e.g., “Assume Step 2 is rate-limiting”).
- Write the elementary step form of the rate law when possible (rate = k[reactants in step]^m).
- If intermediates appear, express them via equilibrium expressions and substitute.
- Label units and justify answers briefly when asked for reasoning.
Study Plans That Actually Work
Consistency beats cramming. A 4-week focused plan before the exam can transform your confidence.
- Week 1: Conceptual foundations — read about rate laws, orders, and basic mechanisms. Do simple initial-rate problems.
- Week 2: Integrated rate laws and graphs — practice identifying orders from plots and calculating half-lives.
- Week 3: Mechanisms and catalysts — work through mechanism-to-rate-law translations and Arrhenius problems.
- Week 4: Mixed practice — timed multiple-choice sections, free-response practice, and targeted review of weak spots.
Personalized practice makes a big difference. If you want structured, one-on-one guidance, Sparkl’s personalized tutoring can help by creating tailored study plans, offering expert tutors who break down tricky mechanisms, and providing AI-driven insights to pinpoint where you lose points. The support is most powerful when it complements regular practice problems and lab-based thinking.
Laboratory Connections: Why Kinetics Isn’t Just Math
Hands-on kinetics experiments reinforce concepts: measuring initial rates, monitoring concentration changes over time (spectrophotometry, conductance, gas volume), and testing the effect of temperature or catalyst. In lab, attention to accurate timing, mixing, and concentration control translates directly into cleaner data and clearer rate laws.
Simple Lab Idea for Practice
- Use the iodine clock reaction to observe how changing reactant concentrations affects time until a visible color change. Time measurements and concentration records can be converted into initial rate data and used to deduce orders.
Real-World Applications That Make Kinetics Cool
Kinetics is everywhere: designing industrial reactors, controlling drug-release rates, understanding atmospheric chemistry, and explaining why food spoils faster at room temperature. Recognizing the broader context helps you remember equations and brings exam examples to life.
Final Tips — Fast Wins for Test Day
- When given initial-rate data, use ratios to find exponents quickly.
- Sketch and label an energy diagram if a question asks about catalysts or activation energy — visuals earn points and clarify reasoning.
- Check units of k: they offer a quick sanity check on your reaction order reasoning.
- Write brief, clear explanations on free-response parts — graders reward concise logic as much as final answers.
- Practice with timed sections and review commonly missed problems to convert weakness into strength.
Parting Thought
Chemical kinetics might seem like a tangle of equations at first, but once you learn to read the rate law as a story — who collides with whom, which step is the bottleneck, and how a catalyst rewrites the script — it becomes a powerful tool. Approach each problem with curiosity: identify the data, map possible mechanisms, and justify every substitution. With steady practice, a few clever strategies, and occasional personalized support from resources like Sparkl’s tutors to guide your weak points, you’ll move from confusion to clarity — and from average to AP-ready.
Good luck, and remember: timing counts — in reactions and in exams. Keep measuring, keep questioning, and enjoy the chemistry.












No Comments
Leave a comment Cancel